May 29, 2024 | Life with MS, News, Progressive, Treatments
On Wednesday 15 May, Multiple Sclerosis NZ raised how critical workforce shortages are impacting patients with Primary Progressive MS (PPMS) access to time sensitive disease modifying therapies. A business case prepared by clinical specialists had been under review for five […]

May 15, 2024 | Advocacy, Funding, Life with MS, Media, News, Press Release, Treatments
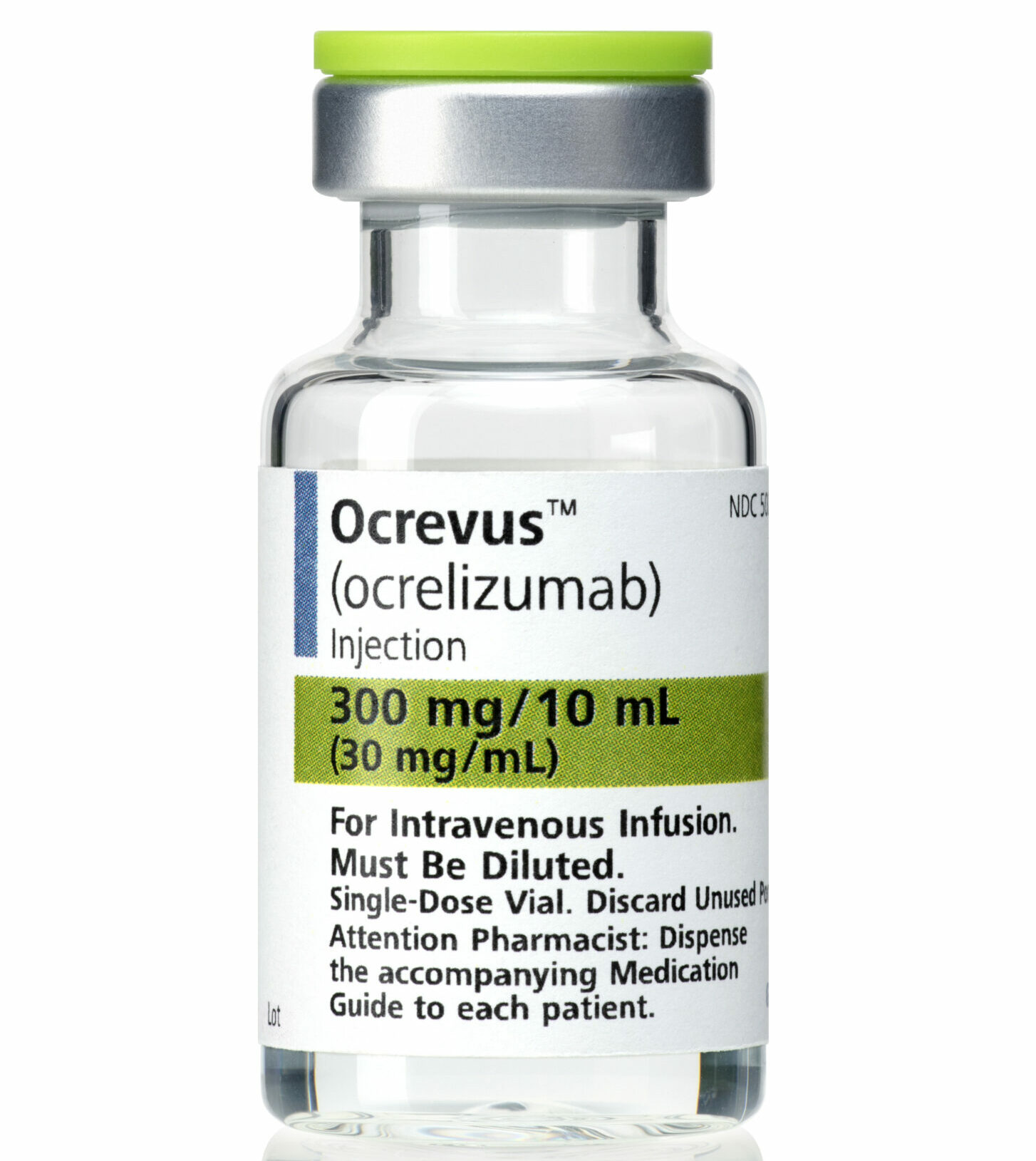
A celebrated Pharmac decision late last year to fund a vital drug for many Multiple Sclerosis patients has fallen flat – with dozens being turned away for treatment due to severely stretched resources across a number of services. Multiple Sclerosis […]

September 4, 2023 | Advocacy, Media, Progressive, Treatments
A seven year battle on behalf of hundreds of people living with the most debilitating form of Multiple Sclerosis has been won – with Pharmac agreeing to fund Aotearoa New Zealand’s first ever treatment for the disease variant from October […]